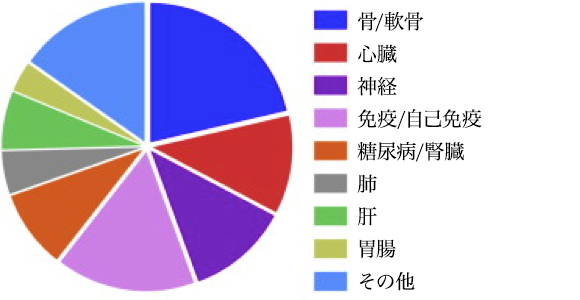
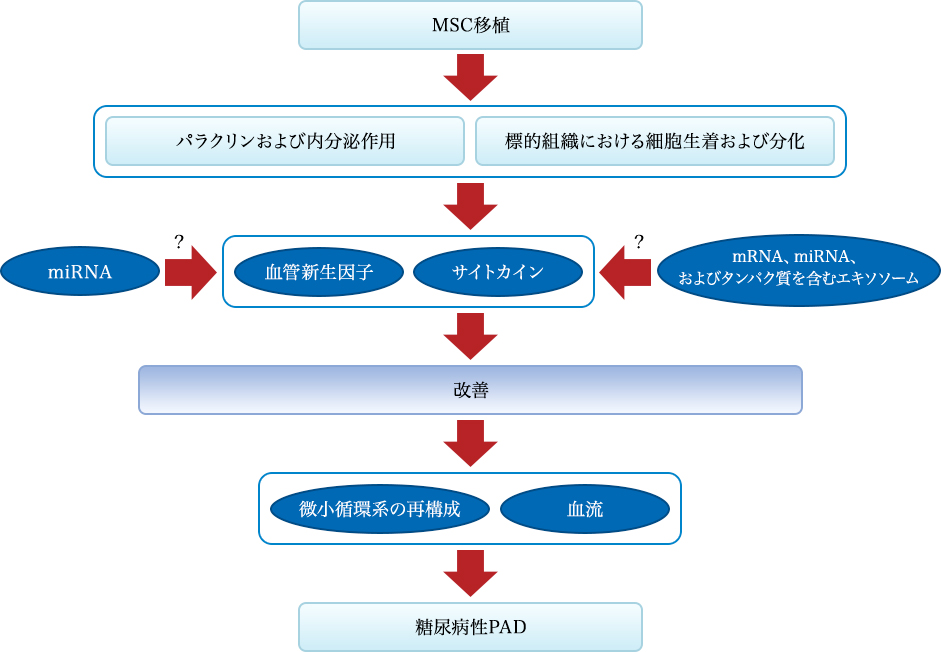
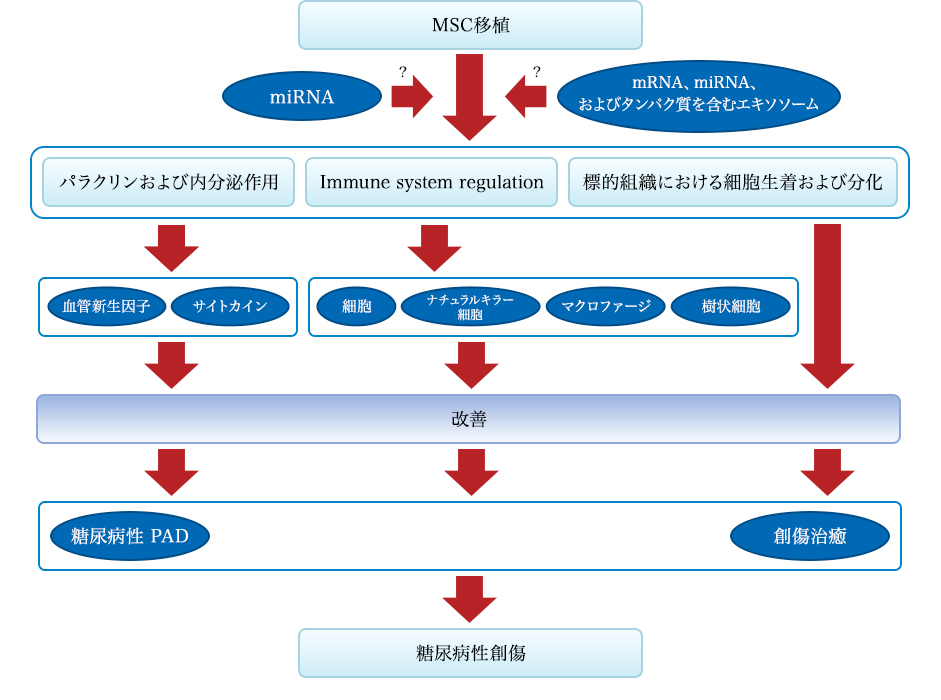
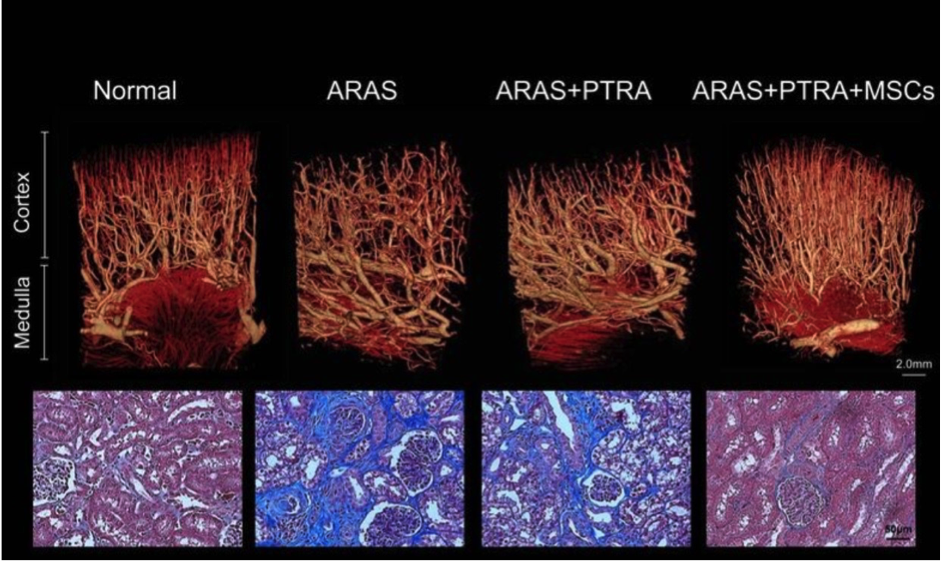
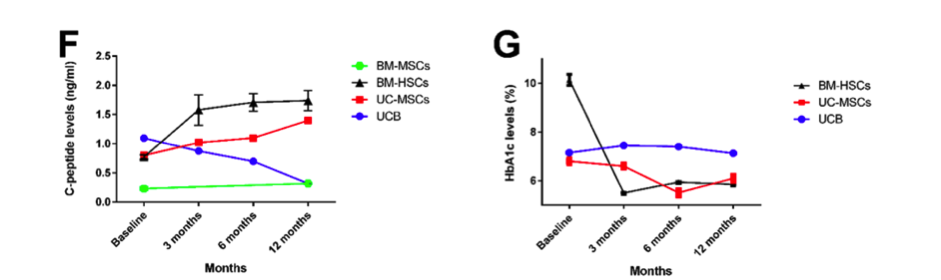
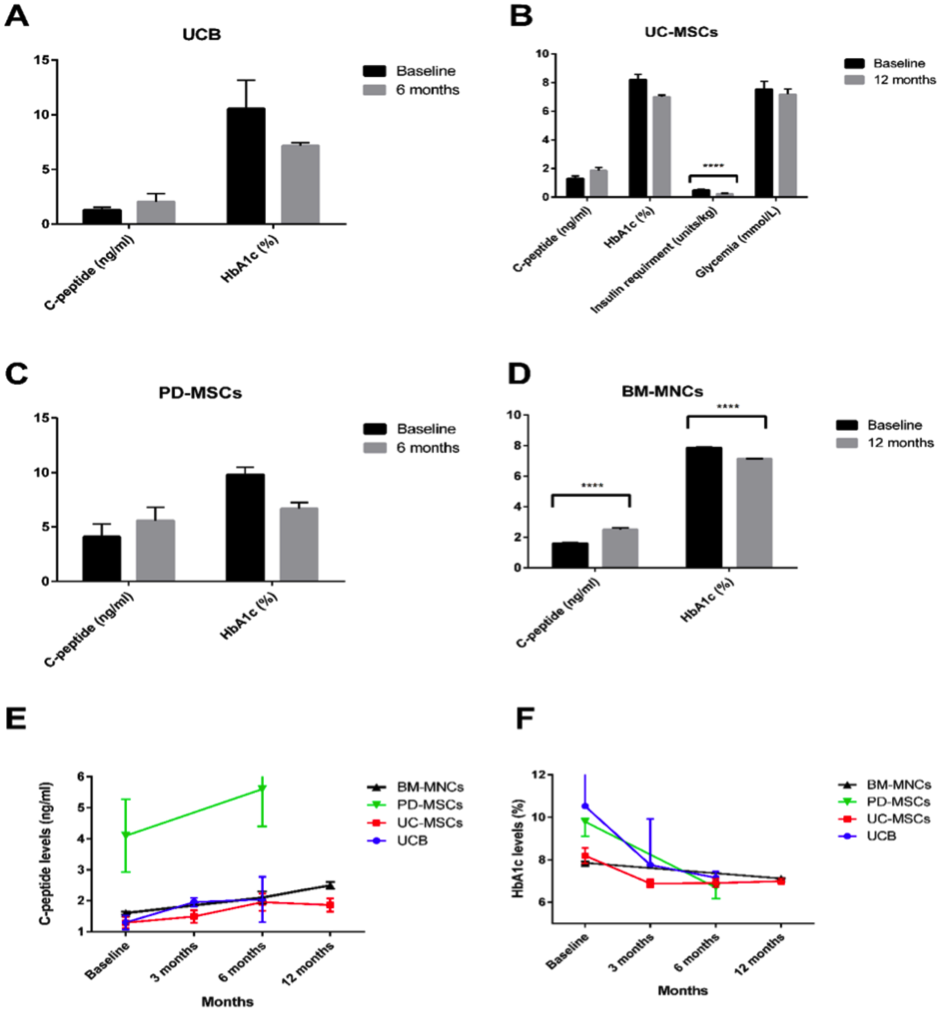
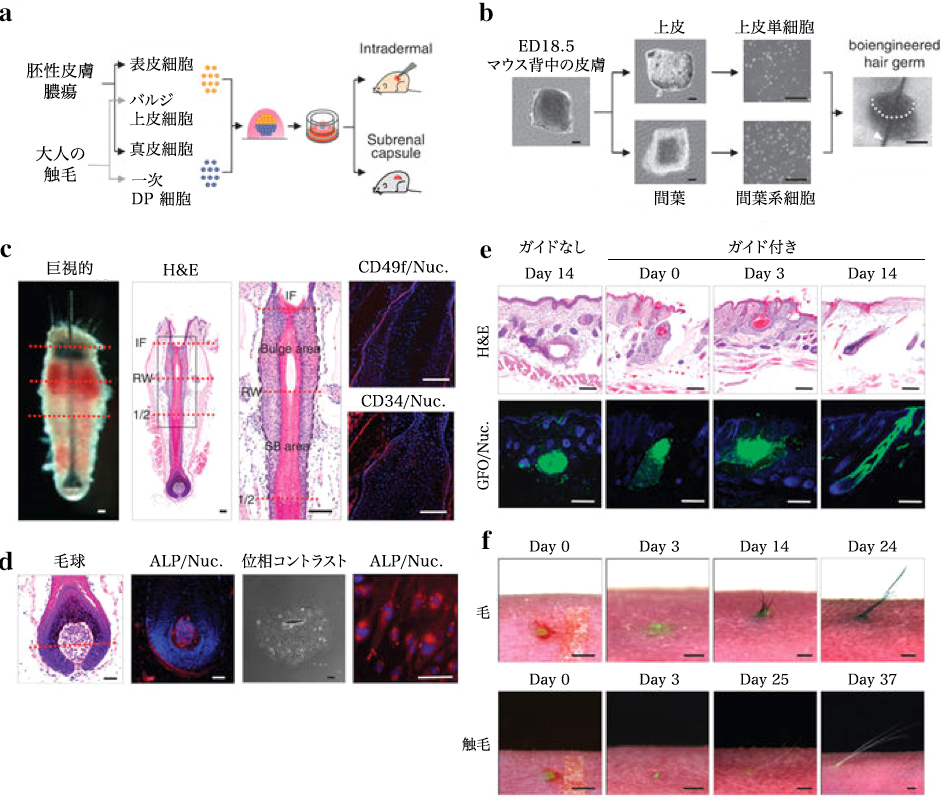
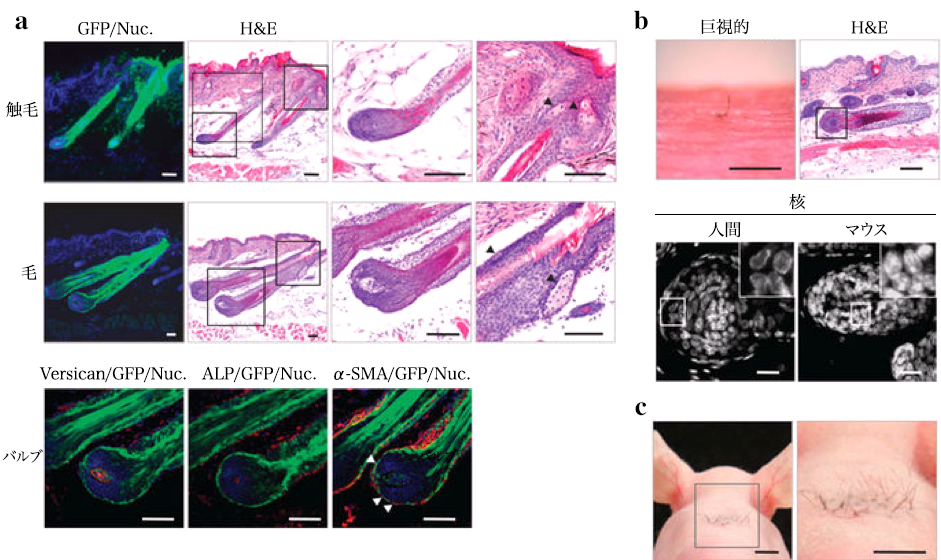
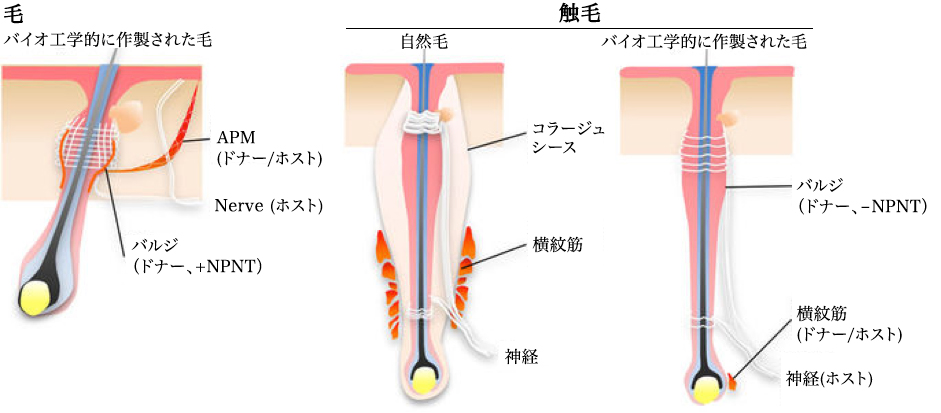
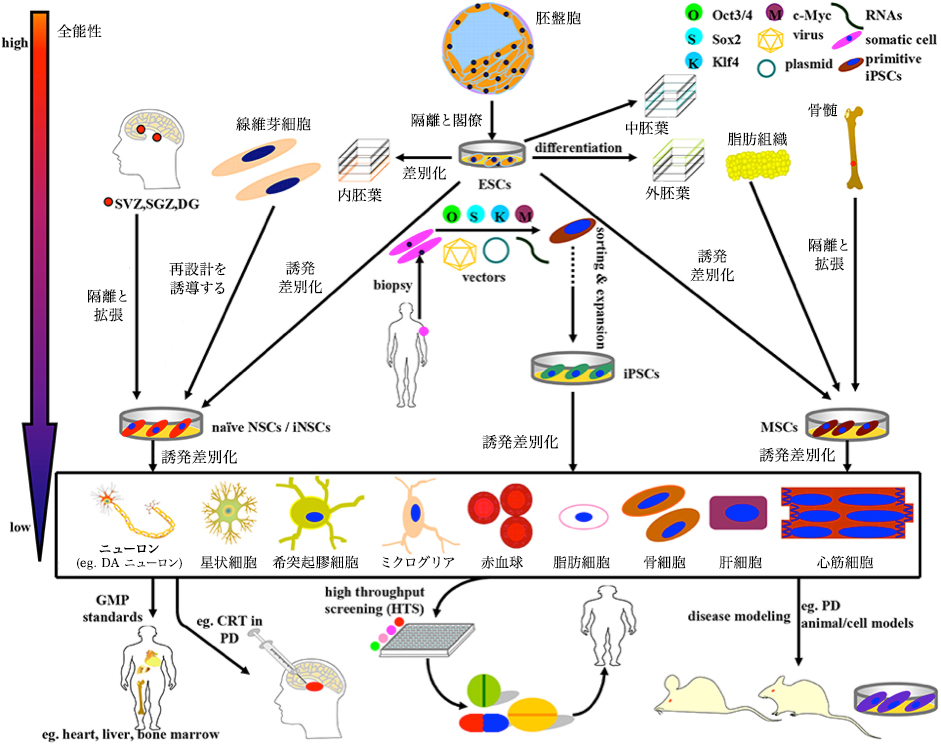
MSC principle
Effect of MSC transplantation on diabetic cardiomyopathy. (A) MSC increases MMP-2 activation, suppresses activation of MMP-9, and attenuates cardiac remodeling. (B) MSCs produce VEGF, IGF-1, AM, HGF and stimulate myogenesis and angiogenesis in damaged myocardium. (C) Via differentiation into cardiomyocytes and vascular endothelial cells, MSC improves myocardial perfusion and myocardial regeneration. Abbreviations: AM, Adrenomedullin; HGF, Hepatocyte growth factor; IGF-1, Insulin-like growth factor-1; MMP, Matrix metalloprotease; MSC, Mesenchymal stem cell; VEGF, Vascular endothelial growth factor
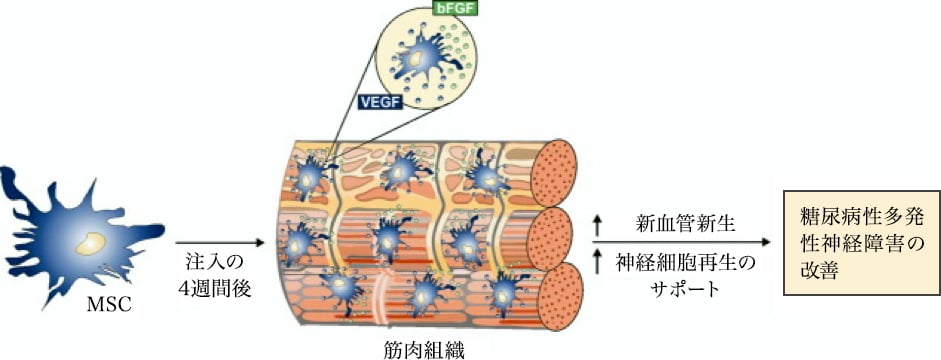
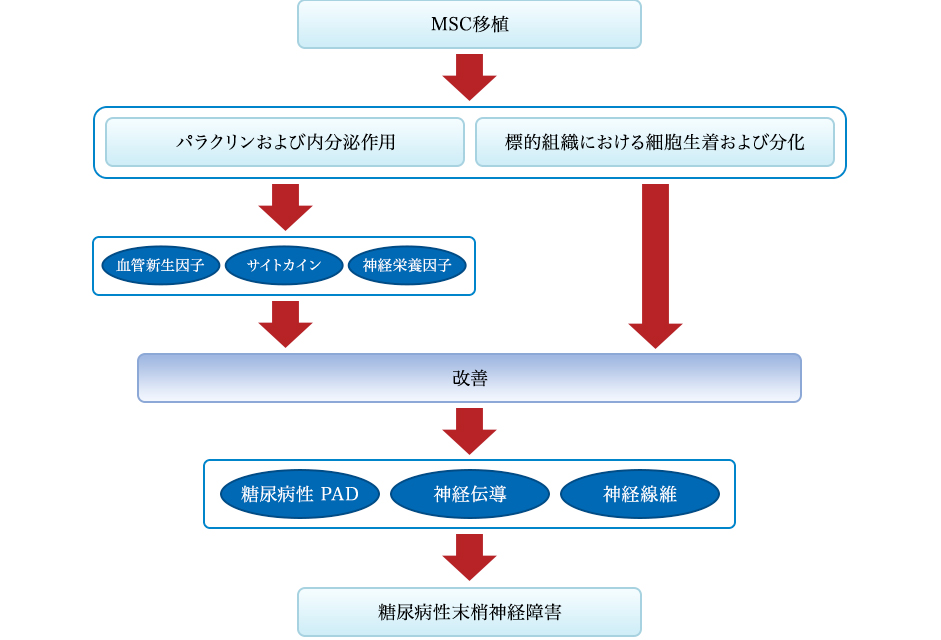
The effect of MSC treatment on diabetic polyneuropathy. Four weeks after intramuscular injection, MSCs deposit in the interstitial space of myofibers through the production of bFGF and VEGF, and induce neovascularization and support regeneration of neuronal cells leading to amelioration of diabetic polyneuropathy. Abbreviations: bFGF, Basic fibroblast growth factor; MSC, Mesenchymal stem cells; VEGF, Vascular endothelial growth factor
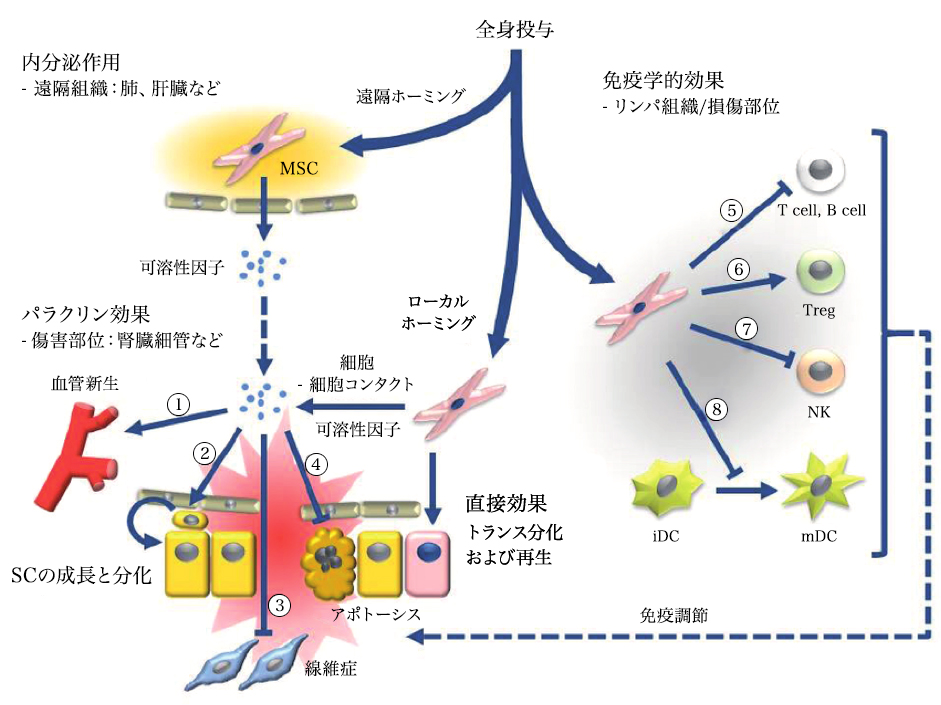
Systemic administration of mesenchymal stem cells can induce endocrine or local paracrine effects, including cell-mediated actions: 1) vascular endothelial growth factor (VEGF), insulin-like growth factor 1 (IGF-1), monocyte chemoattractant protein 1 (MCP1), basic fibroblast growth factor (bFGF), interleukin 6 (IL-6); 2) stem cell proliferation and differentiation: stem cell factor (SCF), leukemia inhibitory factor (LIF), macrophage colony-stimulating factor (MCSF), stromal cell-derived factor 1 (SDF1), angiopoietin 1, and activin A; 3) inhibition of fibrosis: hepatocyte growth factor (HGF), bFGF, and adrenomedullin (ADM); and 4) inhibition of apoptosis: VEGF, HGF, IGF-1, transforming growth factor (TGF)-β, bFGF, granulocyte-macrophage colony-stimulating factor (GM-CSF), activin A, and thrombospondin 1. Immune-mediated effects include (5-8): 5) Suppression of T and B cells: human leukocyte antigen G5 (HLA G5), HGF, inducible nitric oxide synthase (iNOS), indoleamine 2,3-dioxygenase (IDO), prostaglandin E2 (PGE2), bFGF, and TGFβ; 6) Induction of differentiation and proliferation of regulatory T cells (Tregs) through TGFβ expression; 7) Inhibition of natural killer (NK) cells through secretion of IDO, PGE2, and TGFβ; 8) Inhibition of dendritic cell (DC) maturation through secretion of PGE2.
Figure "Stem Cell Res Ther" reproduced by Carrión and Figueroa. 2011 May 11;2(3):23.
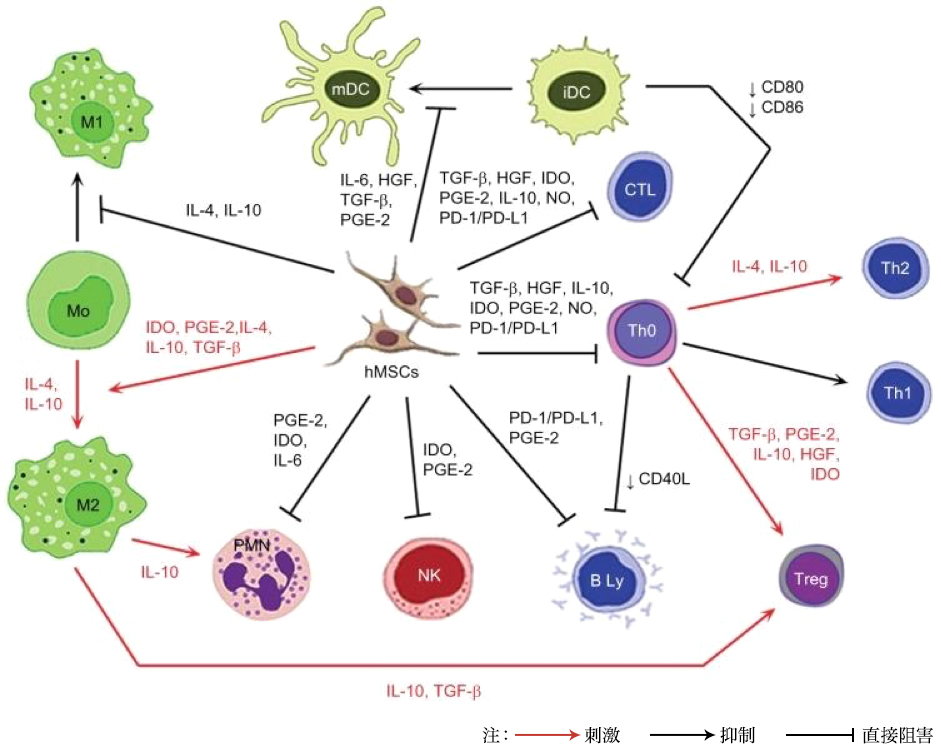
Note: Red arrow: stimulation; black arrow: inhibition; unhooked arrow: direct inhibition.
Abbreviations: iDC, immature dendritic cell; IL, interleukin; HGF, hepatocyte growth factor; TGF-β, transforming growth factor-β; PGE-2, prostaglandin E2; IDO, indoleamine 2,3-dioxygenase; NO, nitric oxide; PD-L1, programmed death ligand 1; hMSC, human mesenchymal stem cell; Treg, T regulatory; Th, T helper; CTL, cytotoxic T cell; mDC, mature dendritic cell; PD-1, programmed cell death protein 1; PMN, polymorphonuclear leukocyte; NK, NK cell
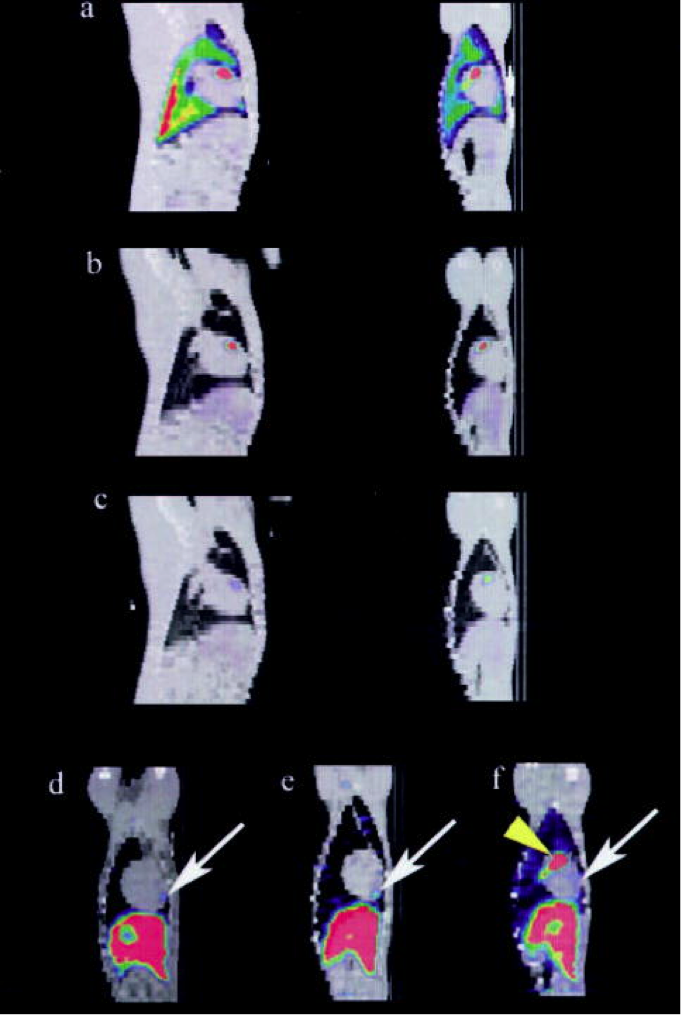
The sagittal plane (left) of the SPECT / CT image fused to day 1 (a), day 2 (b) and day 7 (c) showing local uptake in the anterior ventricular region of the animal’s heart, and this is the coronal plane (right). At the last imaging time (day 5 to 8), the anterior apical area of the MSC uptake (arrow) is shown in three representative animals in the coronal structure map. The vertex distribution ahead of this existed regardless. (Only yellow arrow of f)
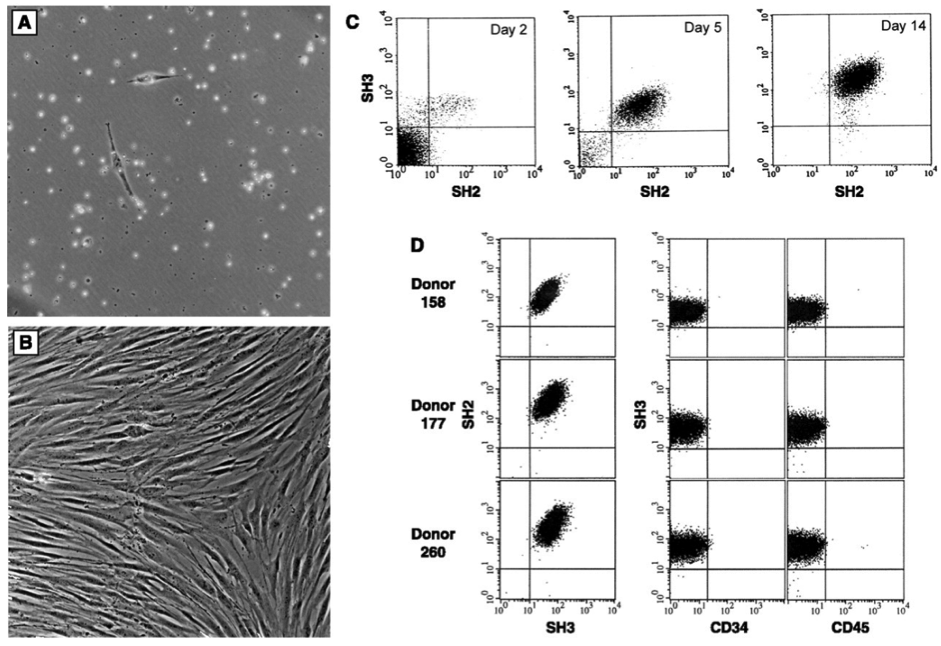
Characteristics of isolated bone marrow stromal cells. Cells are cultured from the bone marrow after density fractionation and are shown as (A) 48 hours after plating and (B) 10 days after plating. (C) Flow cytometry shows enrichment of these cultured cells. Results were obtained on day 2, day 5, and day 14 of culture using antibodies SH2 and SH3 generated against surface markers. (11) On day 14, the cells were 95-99% homogeneous and the reaction to the antigen CD14, CD34 (Becton-Dickinson), or CD45 (Pharmingen) common to hematopoietic cells was negative. (D) Homogeneity and reproducibility of the isolation procedure was demonstrated by flow cytometry