Consult with Professor Helene
You can ask the AI doctor questions by voice.It will answer in your language.

Clicking the button will start a voice conversation
Please talk to me
You can ask questions by clicking the button
You can ask questions like these
TOPICS

- Stem cell therapy
Latest Trends in Stem Cell Therapy for Autoimmune and Inflammatory Diseases
- Stem cell therapy
- regenerative medicine
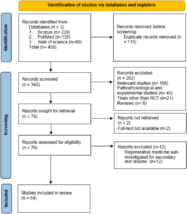
A systematic review of regenerative medicine for skin diseases such as alopecia and vitiligo

- Diabetes
Is chronic fatigue caused by blood sugar imbalances? Its relationship to the body's energy metabolism
Multilingual support(interpreters on staff)

- Japanese
- 50 people
- Chinese
- 18 people
- English
- 16 people
- Minnan language
- 5 people
- Indonesian
- 2 people
- Vietnamese
- 2 people
- Korean
- 2 people
- Mongolian
- 2 people
- Arabic
- 2 people
- Shanghainese
- 1 person
- French
- 1 person
- Tagalog
- 1 person
- Cantonese
- 1 person
- Malay
- 1 person
- German
- 1 person
Latest information
STEMCELL
Stem cell therapy
Stem cells are cells with the ability of re-activating weakened cells and increasing
number of cells that are important to our body function but decreasing every day.
The mechanism of mesenchymal stem cell treatment
Award
Recognized globally for excellence, we hold numerous certifications and
awards, reinforcing trust in our innovative solutions.

Reviewer Certificate
LICENSE
Regenerative medicine provision plans accepted by Ministry of Health, Labor and Welfare
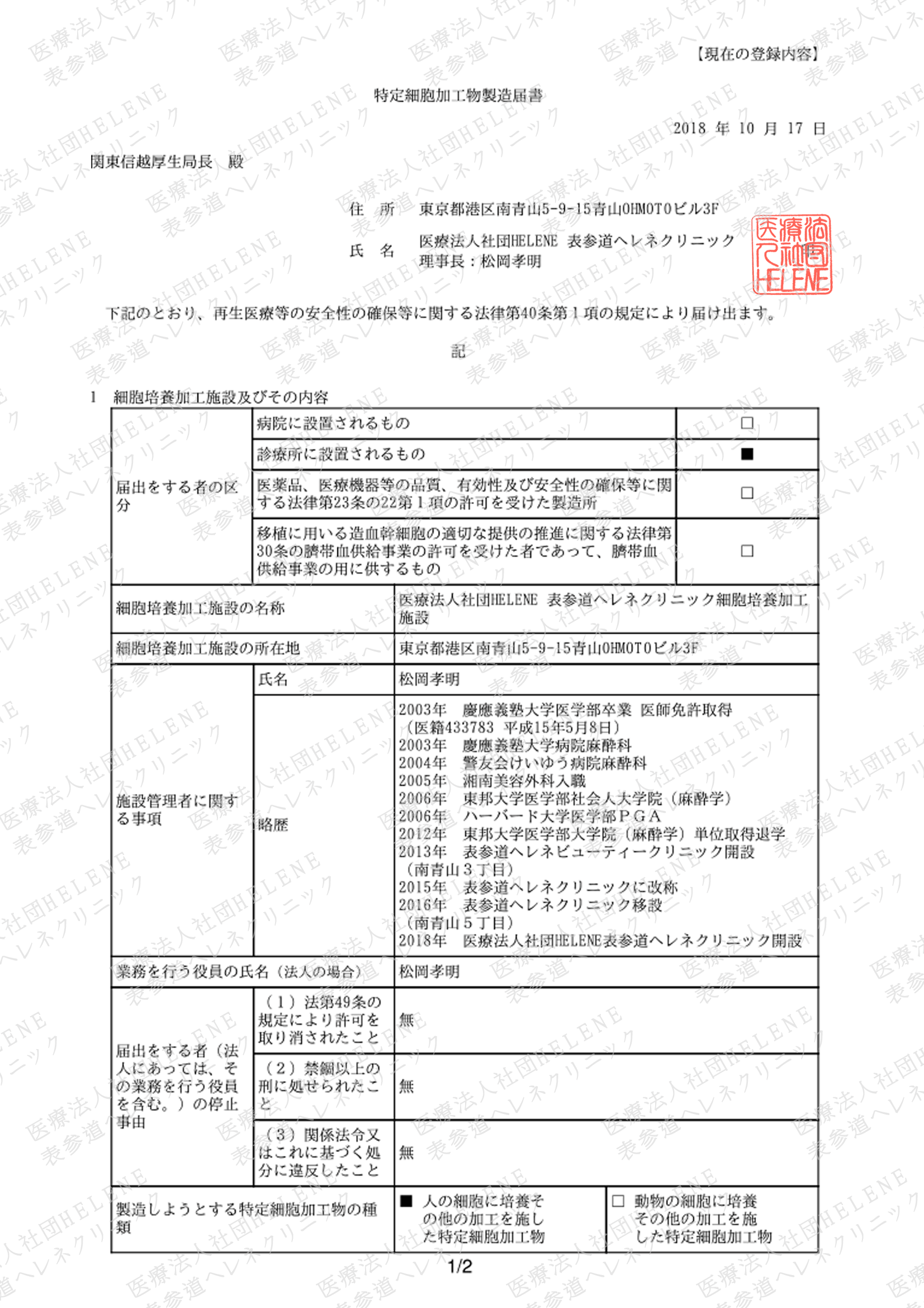
License for cell culturation

License of MSC with old cerebral infarction

Acceptance Certificate for PRP Osteoarthritis

License of MSC with teeth

License of MSC transplantation into joints

License of MSC with MACCE

License of MSC with hair loss

License of MSC in cosmetics

License of MSC and MSC-derived exosomes with immure frailty

License of MSC and MSC-derived exosomes with Type 2 diabetes

Acceptance letter for type 1 diabetes administration

female hormone imbalance Intravenous administration receipt

Receipt for subcutaneous administration to the face

License of Male Erectile Dysfunction

License of Acceptance for Neurodegenerative Disorders

License of Acceptance for Elderly Skin Changes

NK cell administration receipt
Characteristics of HELENE stem cells















FACILITY
Facility of Omotesando cell culture laboratory
Cell Conditioning Facility (CPC) Cell culture is carried out under strict sanitary controls as follows. Inside the clean bench, ISO class 5 (class 100) clean air is supplied, keeping the interior clean. In order to grow and manage cells, cell conditioning facilities (CPCs) must meet very high standards to reduce the number of particles and create a sterile room free of bacteria. In addition, to ensure a high level of safety, we conduct virus tests when cells are received. Furthermore, we have established a cell culture operation management system that incorporates a strict quality control system that includes barcode management (traceability system) and history management of all processes to prevent sample mistakes.
Sự khác biệt về mỡ tùy thuộc vào vị trí thu thập »
GALLERY
Gallery
FAQQ/A
FAQ
BEGGINNER'S GUIDE
Beginner's Guide
SUPPORT