Disease
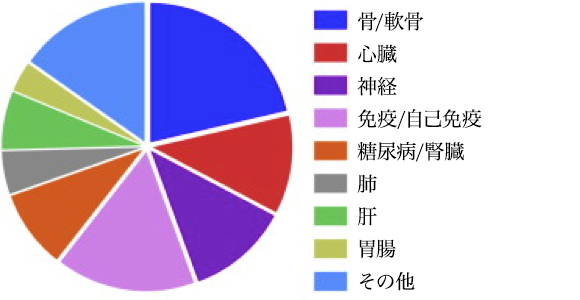
Indications Being Addressed using MSCs in Clinical Trials. Data for 352 registered clinical trials.
Citation// Stem Cell Therapies in Clinical Trials: Progress and Challenges. Trounson, Alan et al. Cell Stem Cell , Volume 17 , Issue 1 , 11 – 22
Disease · Unchanging
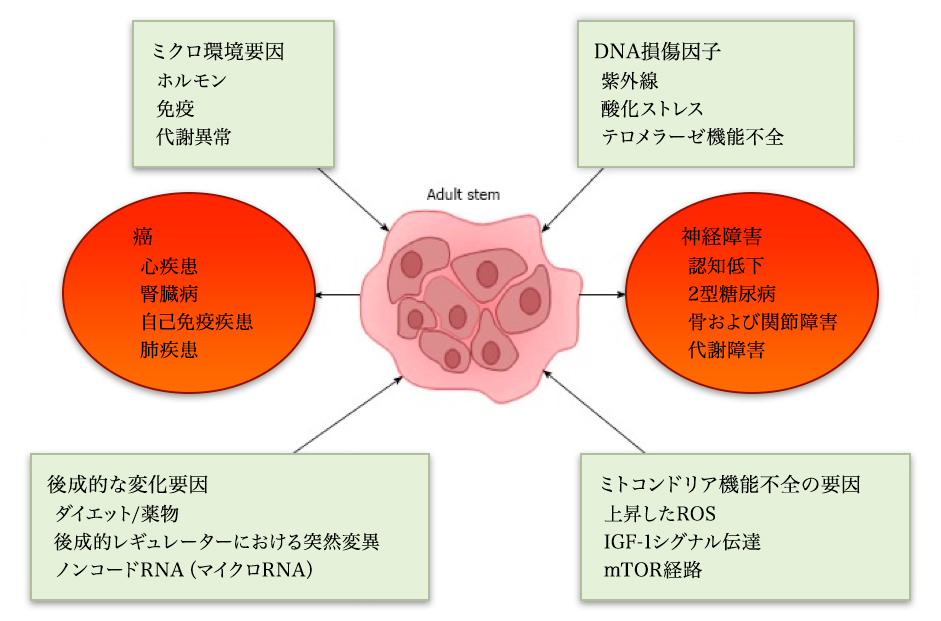
Citation// World J Exp Med. 2017 Feb 20; 7(1): 1–10.Effect of aging on stem cells. Abu Shufian Ishtiaq Ahmed,et al
Disease · Liver / Glycosuria
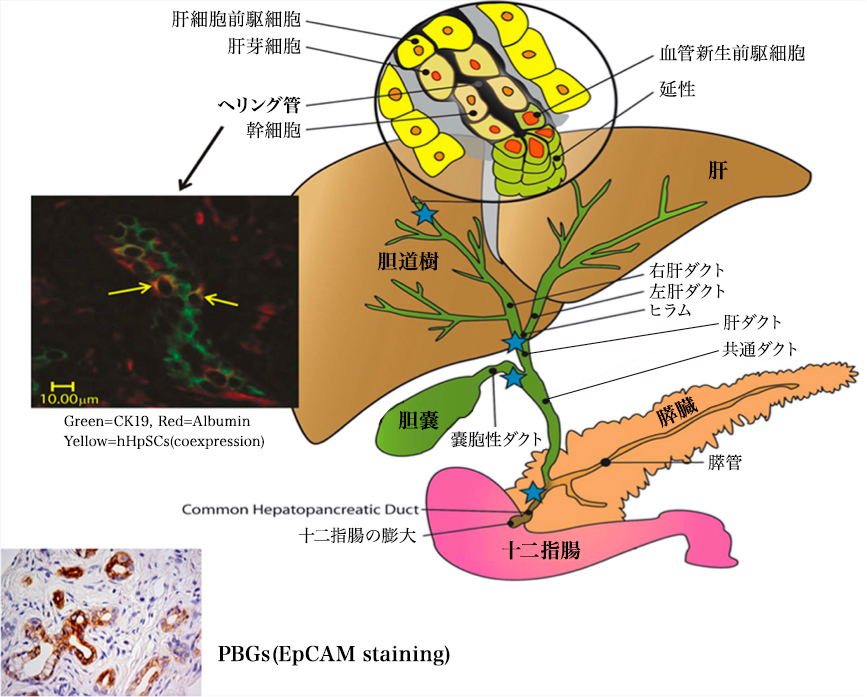
Regenerative medicine is transitioning to clinical programs using stem cell / progenitor cell therapy for repair of damaged organs. Liver and pancreas, organs that share the endodermal stem cell population, biliary stem cells located in the biliary trunk (hBTSC) are briefly described. They are precursors of hepatic stem / progenitor cells in the herring canal and are progenitor cells of the pancreatic duct. They produce a mature line along the radial axis within the bile duct wall and a proximal-distal axis starting at the duodenum and ending at mature cells in the liver or pancreas. Clinical trials have been conducted over the years to evaluate the effects of stem cells transplanted into the hepatic arteries of various liver disease patients (liver stem / progenitor cells from fetal liver). Immunosuppression was not necessary. All control subjects given the given criteria died within 1 year or the liver function declined. Subjects transplanted with 100-150 million liver stem / progenitor cells showed improvement in liver function and survival over several years. Evaluation of the safety and effectiveness of transplantation is still under development. Although stem cell therapy for diabetes using hBTSC is still being studied, it is likely to be done after ongoing preclinical studies. In addition mesenchymal stem cells (MSC) and hematopoietic stem cells (HSC) are used in patients with chronic liver disease or diabetes. MSCs are shown to be effective through paracrine of trophic factors and immune regulators and inefficient lineage restriction to mature parenchymal cells or pancreatic islet cells is limited. The effect of HSC is mainly due to regulation of the immune system.
Stem Cells. 2013 Oct;31(10):2047-60. doi: 10.1002/stem.1457. Concise review: clinical programs of stem cell therapies for liver and pancreas.Lanzoni G1, Oikawa T
Diabetes
Mesenchymal Stem Cells as a Prospective Therapy for the Diabetic Foot
Stem Cells International Volume 2016, Article ID 4612167, 18 pages http://dx.doi.org/10.1155/2016/4612167
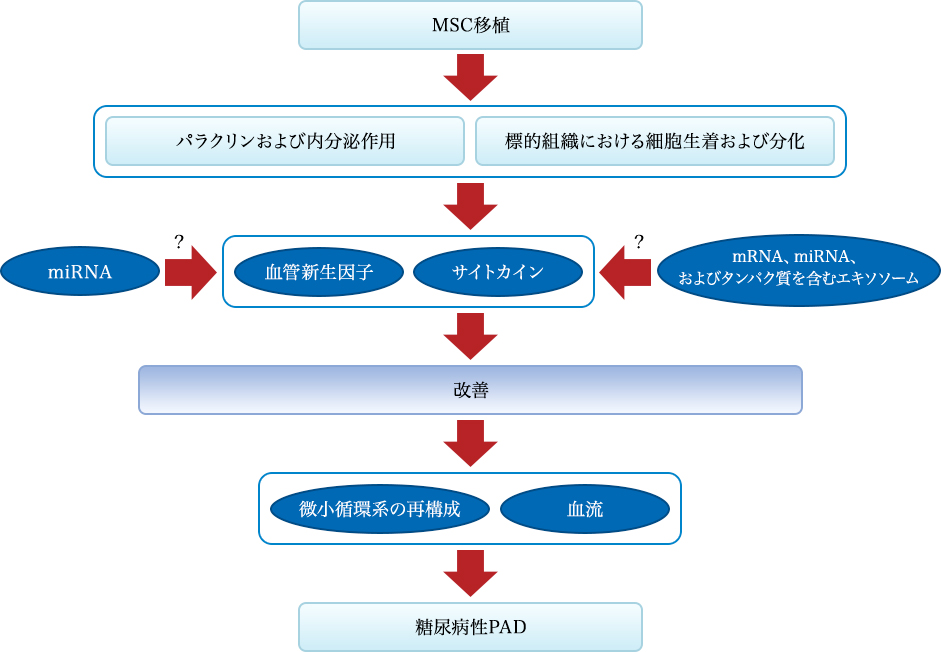
Figure 1: Mechanism of the effect of MSC transplantation on diabetic PAD. Mechanism of recovery effect mediated by stem cell transplantation from two pathways: One is the secretion of angiogenic factors and cytokines, and the other is transplantation and differentiation into tissue constituents of cells. Stem cells can specifically improve the local secretion and expression of angiogenic factors and cytokines, contribute to the reconstruction of the microcirculatory system and improvement of blood flow and islet β-cell function, leading to improvement of diabetic PAD. The stem cells can also differentiate into endothelial cells to achieve recovery of endothelial cell dysfunction. These effects may be related to miRNA and MEX.
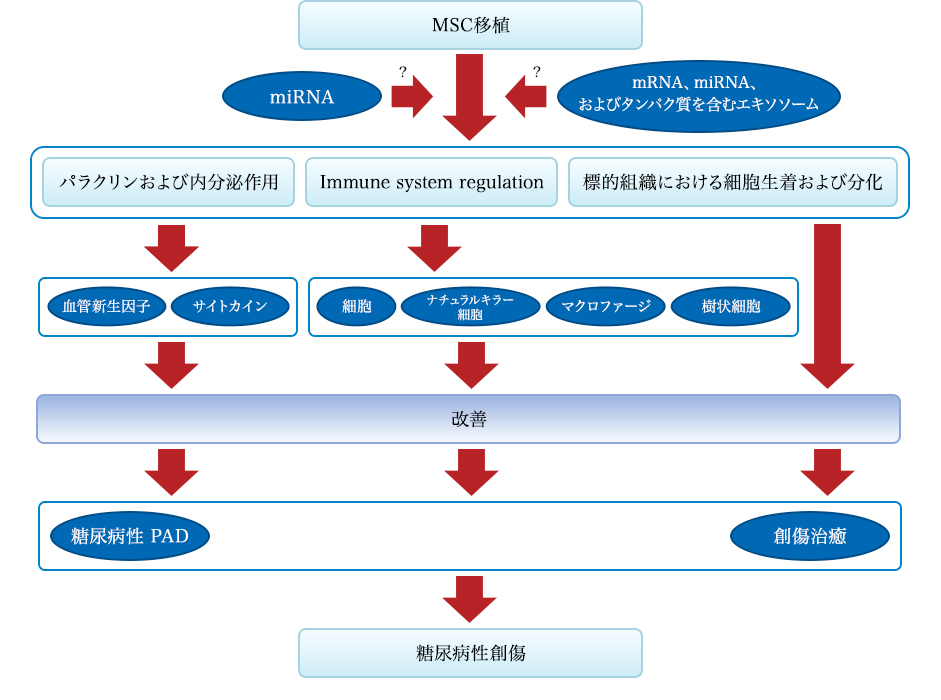
Figure 2: Mechanism of the effect of MSC transplantation on diabetic wounds. Diabetes wound recovery by MSC grafting from three pathways: First is angiogenesis and secretion of factors and cytokines, and second is regulation of the immune system, and third is transplantation and differentiation into tissue constituents of cells is there. Stem cells can specifically improve the local secretion and expression of angiogenic factors and cytokines that contribute to the improvement of diabetic PAD and diabetes. Also, stem cells can regulate the activity of T cells, natural killer cells, macrophages, and dendritic cells and can inhibit infection and inflammatory reactions. In addition, MSCs can differentiate into target tissues and achieve repair. These effects may be related to miRNA and MEX.
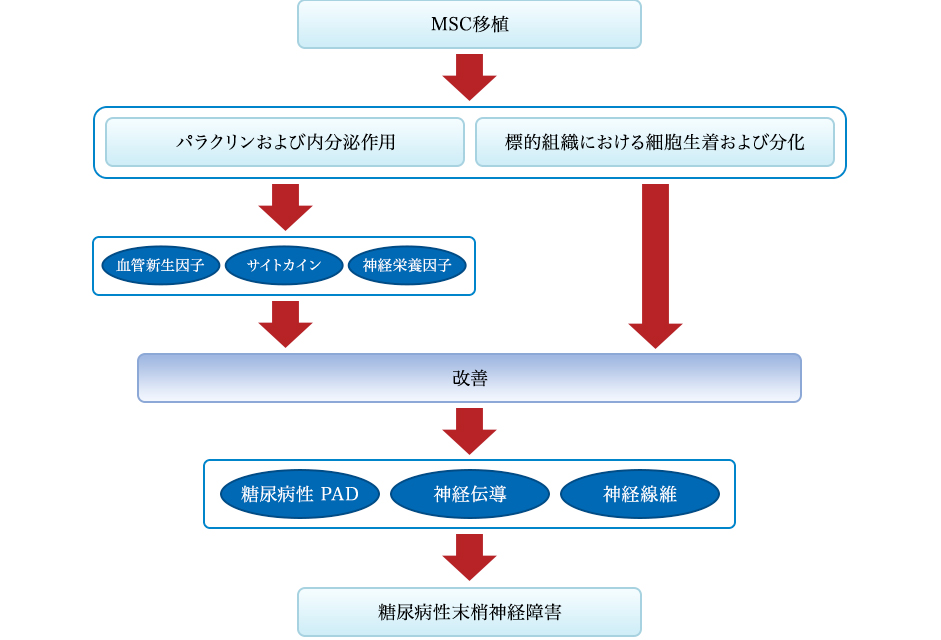
Figure 3: Mechanism of the effect of MSC transplantation on diabetic neuropathy. Mechanism of recovery effect mediated by stem cell transplantation arising from two pathways: One is the secretion of angiogenic factors, cytokines and neurotrophic factors, and the other is transplantation and differentiation into tissue constituents of cells. Stem cells can specifically improve the local secretion and expression of angiogenic factors and cytokines, contribute to the improvement of diabetic PAD and diabetes themselves, leading to amelioration of diabetic neuropathy. Neurotrophic factors can also improve nerve fiber dysfunction and nerve conduction. In addition, stem cells can differentiate into target tissues and achieve repair.
Kidney failure
Stem Cell Research & Therapy 2014, 5:83 http://stemcellres.com/content/5/4/83
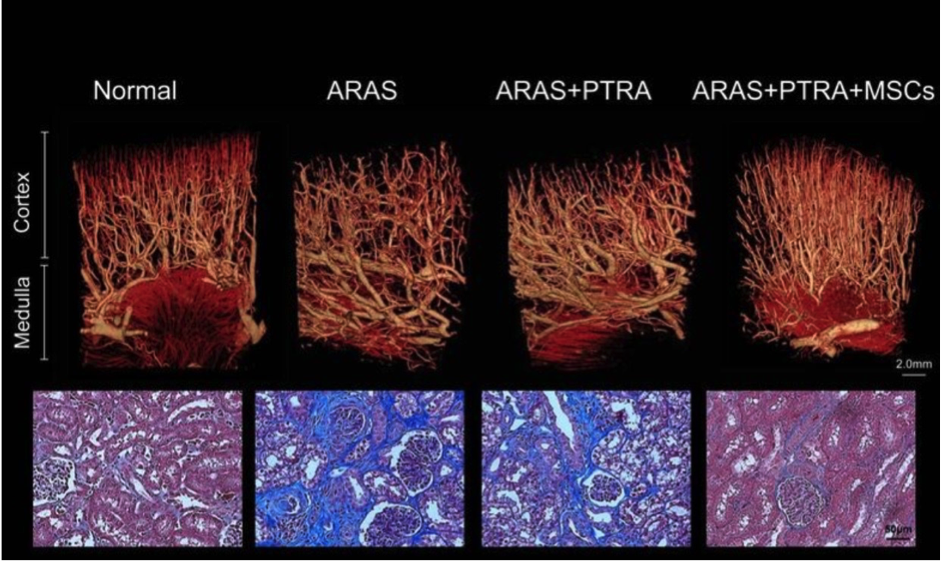
In animals undergoing mesenchymal stem cell therapy, stenosis – renal microvascular loss and fibrosis decreased. Above: Representative microcomputerized tomography of the kidney segment capturing the improved microvascular structure in atherosclerotic renal artery stenosis, pigs who underwent percutaneous transluminal renal angioplasty (PTRA) three dimensional In the image, adrenal infusion of adipose tissue-derived mesenchymal stem cells (MSC) was performed 4 weeks earlier. Bottom: Representative kidney trichrome staining showing a decrease in MSAS fibrosis of ARAS + PTRA + pig (× 40, blue)
Clinical application example of MSC: diabetes
Stem cell transplantation can be a safe and effective treatment for patients with DM. In this series of trials, the best treatment outcome was achieved with D34 + HSC therapy for T1DM, but the worst results with HUCB for T1DM were observed. Diabetic ketoacidosis interferes with the therapeutic effect.
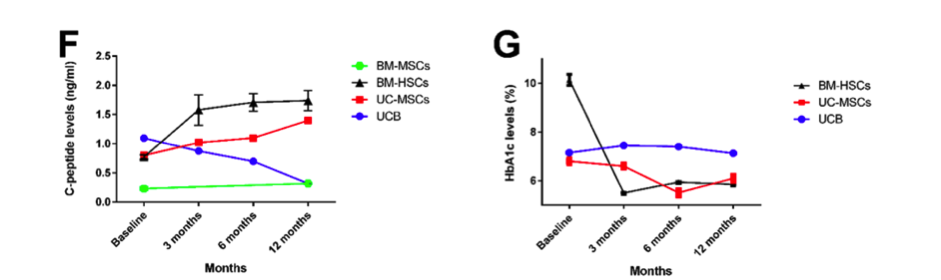
A line graph showing the change in C-peptide and HbA1c levels at baseline, 3 months, 6 months, and 12 months after stem cell treatment in T1DM patients. All data are expressed as mean ± SEM. **** P 0.0001
The outcome for stem cell therapy for T2DM
Stem cell therapy for type 2 DM.
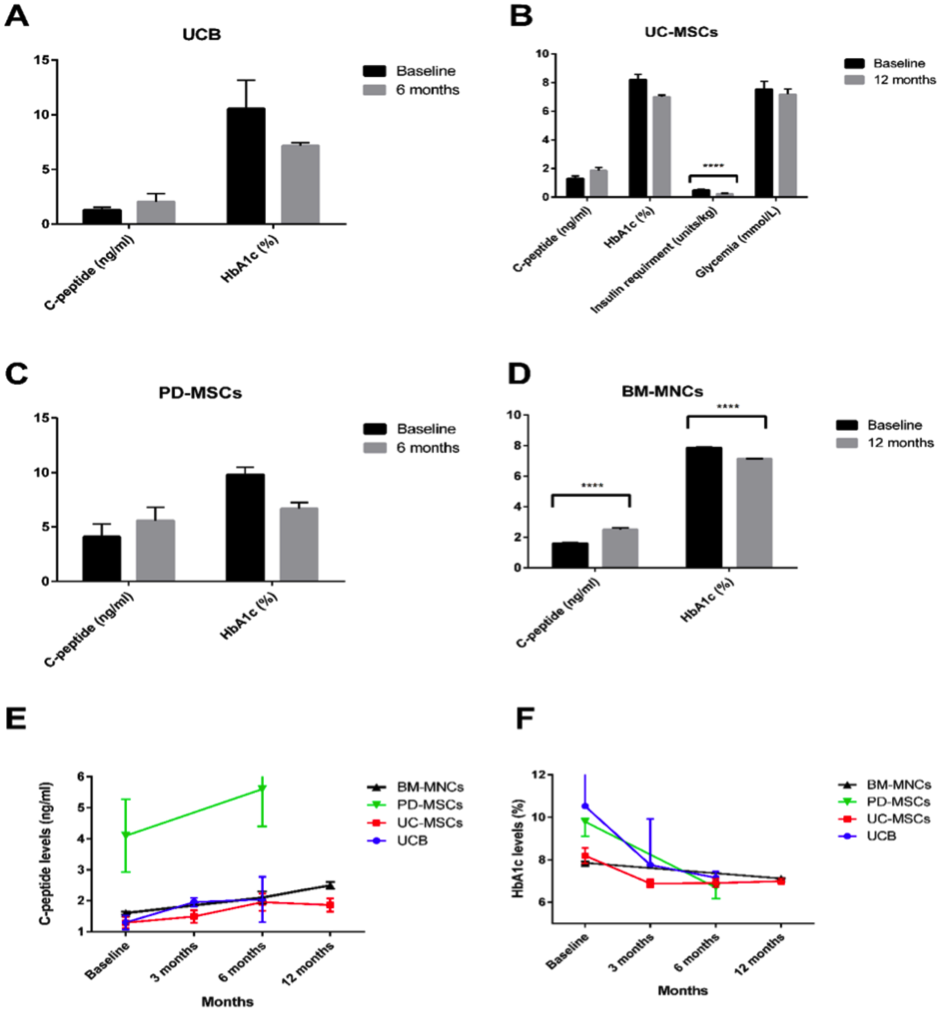
A-D)A bar graph showing the baseline after administration of different types of stem cells, as well as the changes in C – peptide and HbA 1c levels after 12 months. (EF) T2D injected UCB and BM-MNC into the pancreas (n = 3 and n = 107) while intravenously injected with UC-MSC and PD-MSC (n = 22 and n = 10 respectively) A line graph showing the change in C-peptide and HbA1c levels at baseline, 3 months, 6 months, and 12 months after treatment.
Citation// PLoS One. 2016 Apr 13;11(4):e0151938. Clinical Efficacy of Stem Cell Therapy for Diabetes Mellitus: A Meta-Analysis. El-Badawy A, El-Badri N.
Disease · Hair follicle
Fully functional hair follicle regeneration through the rearrangement of stem cells and their niches.
Toyoshima KE1, Asakawa K, Ishibashi N, Toki H, Ogawa M, Hasegawa T, Irié T, Tachikawa T, Sato A, Takeda A, Tsuji T.
Overview
Organ replacement regenerative medicine promises to enable the replacement of organs damaged by disease, injury, or aging in the foreseeable future. Here, we demonstrate fully functional organ regeneration via intradermal transplantation of bioengineered bone and blastoderm. The blastoderm and ovule are reconstituted with embryonic skin-derived cells and adult stem cell field-derived cells, respectively. The bioengineered hair follicles develop the correct architecture and form appropriate connections with surrounding host tissues, such as the epidermis, hindlimb muscle, and nerve fibers. The bioengineered hair follicles also exhibit restored hair cycle and hair head formation through reorganization of hair follicle stem cells and their niches. This study therefore reveals the potential of adult tissue-derived hair follicle stem cells as a bioengineered organ replacement therapy.
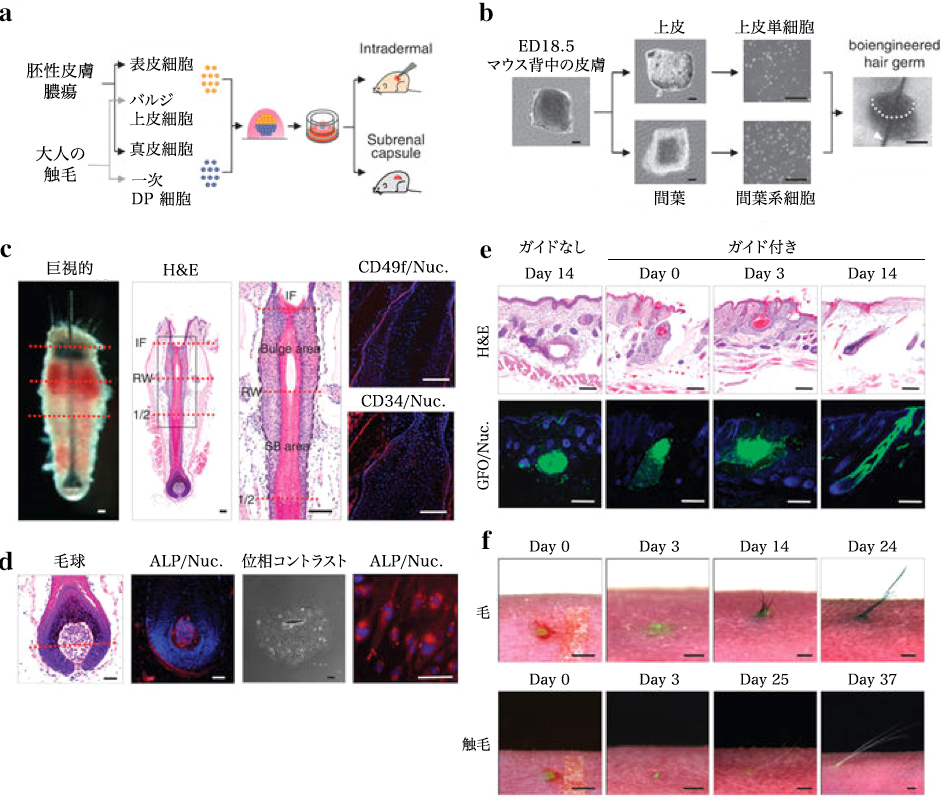
(a) Schematic of the method used for making and transplanting bioengineered hair follicle embryos. (b) Phase difference images of dorsal skin, tissues, dissociated single cells and bioengineered hair follicle embryos of mouse embryos reconstructed using the organ germ method using nylon thread (arrow head). Scale, 200 μm. (c) Histological analysis of isolated tentacles from adult mice. Macroscopic and H & E staining bristles are shown on the left two panels. The dashed line (red) by macro morphological observation (left) and H & E staining (right) shows the interface of bulge and SB region. The box area on the left panel is stained with H & E to show the bulge and the SB area is shown on the higher panel in the higher panel. The bulge region was immunostained with anti-CD49f (red, left) and anti-CD34 (red, middle) antibody and Hoechst 33258 dye (blue). The black dashed line at high magnification H & E indicates the interface of the epithelium of the follicle. IF, funnel; RW, annular body; half of hair follicle. Scale, 100 μm. (d) Histological and ALP analysis of initial regeneration of tactile valve area and DP cells. Hair bulbs (2 left) and cultured DP cells (2 right) were analyzed by ALP enzyme staining. The red dotted line shows Auber’s line. Scale “, 100 μm. (e) Longitudinal section of bioengineered hair during the eruption and growth process, mediated by the inter-epithelial tissue-interlocking plastic device (guided). Corresponding items are (without guide) skin It is shown as cystogenesis with bioengineered hair follicles 14 days after transplantation H & E staining (top) of bioengineered hair follicles on days 0, 3 and 14 post- Fracture microscope (bottom) Scale, 100 μm (f) Healing of wounds immediately after implantation (left), day 3 (center) on day 0, ejection of hair shafts on days 14 and 37 , And macrostructural observation of grown hair during development and growth of bioengineered chest (upper) and spleen (lower), including growth Scale, 1.0 mm.
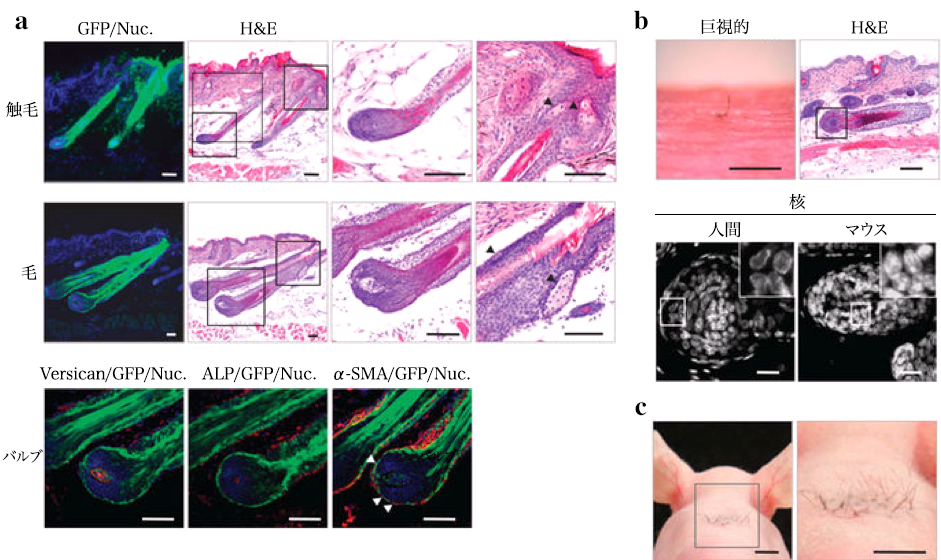
(a) Histological and immunohistochemical analysis of bioengineered hair (upper) and tentacular (central) vesicles. The box area of the low magnification H & E panel is shown at higher magnification in the right panel. Arrows indicate sebaceous glands. Scale, 100μm. Bioengineered follicle hair bulbs were immunostained with anti-versican (lower left) and α-SMA (arrow head, lower right) antibodies and also enzymatically stained for ALP. (Lower center) scale, 50 μm. (b) Bioengineered human hair produced by transplantation of biologically treated hair follicle embryos were reconstituted by intact DP of bulge derived epithelial cells and human scalp follicles. Human hair by bioengineering was photographed (microscopic examination) on day 21 after transplantation and analyzed by H & E staining. According to nuclear morphological characteristics (right panel), species identification of bioengineered hair follicles was analyzed. The enclosed area of the inset is high magnification. Scale, microscope 500 μm, H & E 100 μm, nuclear dyeing 20 μm. (c) Dense intradermal transplantation of bioengineered follicular pathogens. A total of 28 independent bioengineered hair follicle germs were transplanted into the uterine cervical skin of the mouse and showed high density hair growth on day 21 after transplantation. Scale, 5 mm.
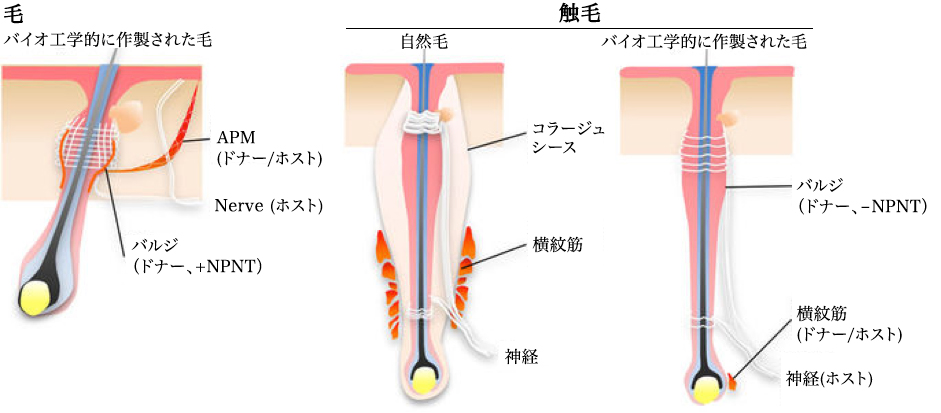
Bioengineered hairs and tentacles were connected with other tissues such as nerve fibers, retractor muscles and striatum muscles derived from host or donor cells. The bioengineered hair bound smooth muscle as a result of regeneration of the NPNT-expressing bulge region, similar to the natural mass. Neither NPNT expression nor smooth muscle binding was detected in the bioengineered hair bulge region.
Quote/Fully functional hair follicle regeneration through the rearrangement of stem cells and their niches. Koh-ei Toyoshima, Kyosuke Asakawa, Naoko Ishibashi, Hiroshi Toki, Miho Ogawa, Tomoko Hasegawa, Tarou Irié, Tetsuhiko Tachikawa, Akio Sato, Akira Takeda & Takashi Tsuji. Nature Communications 3, Article number: 784 (2012)
doi:10.1038/ncomms1784
Other diseases · Parkinson
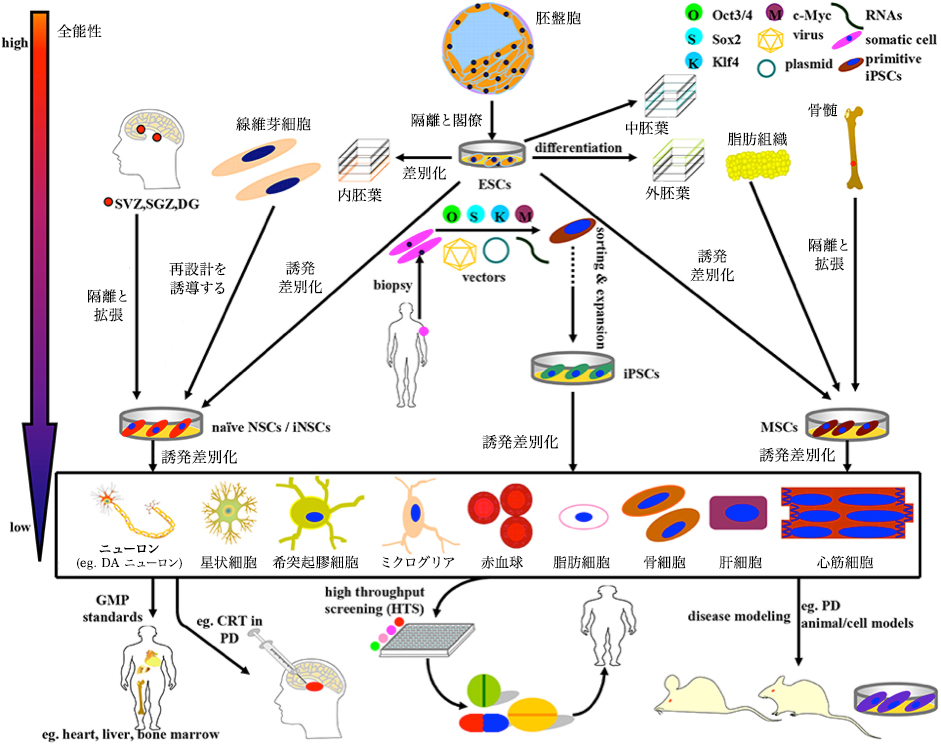
Schematic of induction, differentiation and application of stem cells currently available for PD research and therapy. The above stem cells can be divided into four categories: ESC, NSC, MSC, and iPSC, with gradually decreasing differentiation totipotency. (1) ESC originating mainly from the blastocyst inside the blastocyst can simultaneously differentiate into endoderm, mesoderm and ectoderm under normal circumstances. In some cases ESC can also be induced to differentiate into NSC and MSC. (2) NSC isolated directly from specific brain niche or reprogrammed from fibroblasts can perform neural lineage differentiation into neurons and almost all glial cells. (3) MSC is mainly derived from mesenchymal tissue and can differentiate into almost all cells of mesoderm origin. Remarkably, MSC can be induced to differentiate into DA neurons even under certain combinations of induction protocols. (4) iPSCs that can be re-differentiated from adult human somatic cells (such as fibroblasts) by introducing OSKM (Oct 3/4, Sox 2, Klf 4, and c-Myc) It is a promising source of stem cells to have. Based on the GMP standard, the stem cells and terminally differentiated cells described above can be further sorted, purified and expanded for application to disease model, drug screening, and CRT practice. For example, ESC, MSC, NSC, and DA neurons are used below. (I) Preparation of PD model (ii) Potential drug screening; (iii) CRT treatment of PD Front. Aging Neurosci., 31 May 2016. A Compendium of Preparation and Application of Stem Cells in Parkinson’s Disease: Current Status and Future Prospects. Yan Shen, Jinsha Huang